Key Takeaways
- Efficacy: Both treatments produce strong, rapid relief from treatment-resistant depression. Head-to-head meta-analyses suggest IV ketamine may achieve slightly higher response and remission rates, while Spravato shows the strongest long-term relapse-prevention data.[1][2]
- Cost: Commercial insurance often covers Spravato (esketamine) for eligible patients with treatment-resistant depression, while IV ketamine infusions are typically self-pay at $400–$600 per session for uninsured patients.[3]
- Administration: Patients receive IV ketamine through a clinician-administered intravenous drip with flexible dosing. Spravato is a self-administered, fixed-dose nasal spray taken in-clinic under supervision.
- Suicidality: Both treatments can reduce suicidal thoughts within hours of the first session, which is one reason ketamine-based therapies are used for acute depressive crises.[4]
At-a-Glance Summary: Which is Better?
Neither IV ketamine nor Spravato works better for everyone. IV ketamine often provides more flexible dosing, faster onset, and stronger response or remission outcomes in some comparative analyses. Spravato offers a standardized, FDA-approved, insurance-accessible pathway with strong relapse-prevention data and required clinical monitoring.[1][2]
The better option depends on depression severity, suicidal thoughts, prior treatment history, cost, insurance coverage, and how a patient responds during the first few weeks of Spravato or the first IV ketamine series.
At Avesta Ketamine and Wellness, patients work with an experienced clinical team to determine whether IV ketamine or Spravato is the better fit for their symptoms, treatment goals, and long-term mental health needs.
Spravato vs Ketamine: At-a-Glance Comparison
| Factor | Esketamine (Spravato®) | Ketamine (IV infusion) |
|---|---|---|
| Chemical form | S-enantiomer only | Racemic (R + S enantiomers) |
| FDA approval | Approved for treatment-resistant depression (2019) | Approved as anesthetic; used off-label for depression |
| Route | Nasal spray, self-administered under supervision | IV infusion, clinician-administered |
| Session length | ~2 hours (including monitoring) | 45–60 minutes |
| Typical schedule | 2x/week for 4 weeks, then weekly | 6 infusions over 2–3 weeks, then maintenance |
| Insurance coverage | Often covered after 2 failed antidepressants | Usually out-of-pocket; some clinics in-network |
| Out-of-pocket cost (uninsured) | $800–$1,200 per session | $400–$600 per session |
| Approved for ages | 18+ only | Used in teens off-label |
| Other indications | MDD with TRD only | PTSD, OCD, anxiety, chronic pain, SUD |
Is Spravato Ketamine Therapy? Understanding the Difference
Spravato is a form of ketamine therapy, but it is not identical to IV ketamine treatment in substance or protocol.[5]
- Spravato contains esketamine, an FDA-approved ketamine-derived medication developed specifically for depression and delivered as a nasal spray.
- IV ketamine uses racemic ketamine delivered directly into the bloodstream through an infusion.
Both treatments act on the brain’s glutamate system, which helps regulate mood and the formation of new connections.[6] However, dosing and frequency differ:
- Spravato is administered according to standardized dosing (56 mg or 84 mg) in a certified clinical setting for at least 8 weeks.
- IV ketamine allows the clinical team to adjust the dose, infusion rate, and number of sessions based on the patient’s response.
For some patients, that flexibility makes IV ketamine the better fit. For others, Spravato’s FDA-approved structure and insurance pathway make treatment more accessible.
Chemical Differences

At a chemical level, esketamine is one half of the ketamine molecule — the S-form. Ketamine itself is a racemic mixture containing both R-ketamine (arketamine) and S-ketamine (esketamine).[7] In preclinical studies, esketamine binds NMDA receptors with 2–3x the affinity of R-ketamine,[8] but more recent animal research suggests R-ketamine may produce more potent and longer-lasting antidepressant effects.[9]
These chemical variations affect dosing control and how the body processes each medication. Patients do not need to memorize the molecular terms — the takeaway is that Spravato and IV ketamine are related but clinically distinct treatments, so their effects, dosing, insurance coverage, and patient fit can differ.
How the Experience Can Differ
Spravato and IV ketamine both create temporary dissociation, which can feel like a dreamlike, detached, or psychedelic-like state.
IV ketamine can feel more immersive. IV ketamine enters the bloodstream directly and steadily through the infusion. Many patients describe the experience as deeper, more internal, or more psychedelic than Spravato, especially as the dose increases.
Spravato effects may feel more gradual or contained. Spravato absorbs through the nasal passages, so the experience can feel less direct than an infusion. Some patients still have a strong dissociative experience, but others describe it as lighter, more uneven, or easier to stay oriented through.
The timing can feel different. IV ketamine effects develop soon after the infusion begins and fade rapidly as the infusion ends. Spravato effects build after dosing and then gradually taper across the required 2-hour monitoring period.
How Spravato vs. Ketamine Works for Depression
IV ketamine and Spravato work for depression through the same core mechanism: both rapidly increase glutamate activity in the brain. Glutamate is the brain’s main excitatory neurotransmitter, meaning it helps brain cells communicate, adapt, and form new connections.[10]
This glutamate surge matters because chronic depression weakens communication between mood-related brain circuits.[11] When those circuits become less flexible, the brain has a harder time adjusting its emotional responses to daily life. Neutral experiences may begin to feel threatening, small setbacks can feel overwhelming, and motivation becomes harder to access.
Ketamine-based treatments help the brain’s regions reconnect. Spravato and IV ketamine improve flexibility by increasing activity in glutamate pathways that support synaptic growth — the tiny connection points where brain cells pass messages to each other.
Stronger connections help patients better access and manage a range of emotions. The brain may begin responding with more relief, perspective, motivation, or steadiness. Researchers call this repair-and-adaptation process neuroplasticity, and it helps explain why some patients notice meaningful symptom improvement within days or weeks of starting IV ketamine or Spravato.
What to Expect: Spravato Treatments vs IV Ketamine Infusions
Spravato and IV ketamine are both in-clinic treatments that require monitoring and multiple sessions for best results. The protocols differ in schedule, appointment length, dosing, and maintenance planning.
IV Ketamine Infusion Protocol
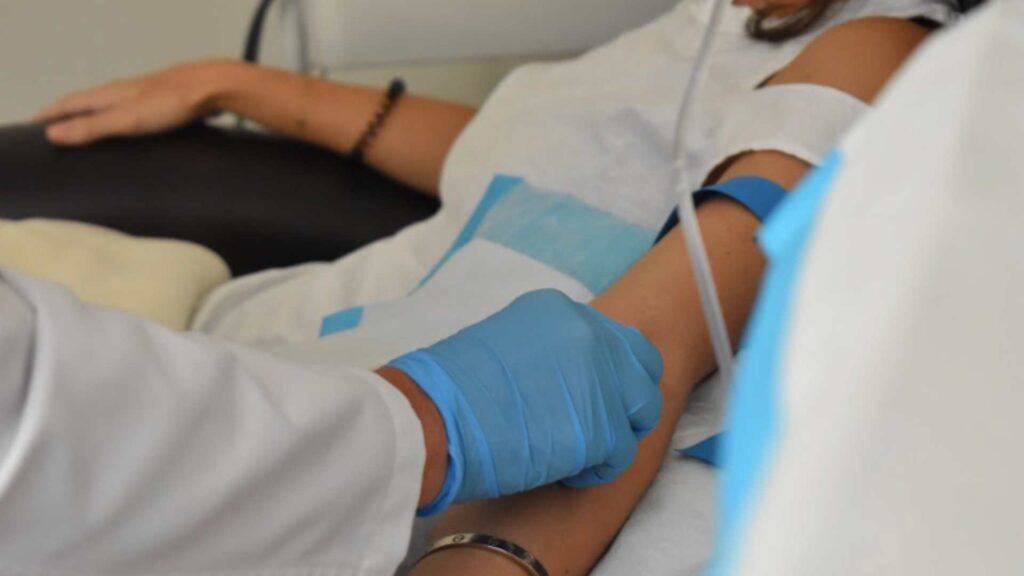
The typical introductory protocol for IV ketamine is a series of six infusions over two to three weeks, with significant room for customization.
- Administration and dosing: A trained clinical provider administers ketamine through an intravenous (IV) line. Dosing is typically weight-based, and the clinical team can adjust the dose or infusion rate based on the patient’s response, comfort, and safety needs.
- Appointment length and recovery: Each infusion lasts about 45–60 minutes, followed by recovery time before the patient leaves the clinic. Patients need someone to drive them home and should not drive for the rest of the day.
- Maintenance planning: After the first series, some patients return for booster infusions when symptoms recur. Timing varies by patient.
Spravato Treatment Protocol
Spravato follows a labeled dosing schedule that begins with twice-weekly treatments for 4 weeks, then tapers to once weekly for the next 4 weeks if symptoms improve.
- Administration and dosing: Patients self-administer Spravato nasal spray in the clinic under clinical supervision. The starting dose is typically 56 mg on day one, then 56 mg or 84 mg depending on response.
- Treatment schedule: Spravato is given twice weekly during weeks 1–4, once weekly during weeks 5–8, and once weekly or every two weeks thereafter based on patient needs.
- Appointment length and recovery: Each Spravato visit includes dosing plus at least two hours of observation before the patient leaves. Patients need a ride home and should not drive until the next day after a full night of sleep.
Side Effects
Esketamine and ketamine produce nearly identical short-term side effects, with some differences in how people experience them.
Both can cause dissociation (a sense of detachment from reality), disorientation, nausea or vomiting, temporary increases in blood pressure and heart rate, and anxiety during the session. Research suggests esketamine causes less drowsiness[12] and cognitive impairment after dosing, while IV ketamine more often produces a mood-elevating,[13] mildly euphoric experience.
Side effects from both typically resolve within a few hours, and serious adverse events are rare when administered in a supervised clinical setting.
Research: Spravato vs Ketamine for Depression
Clinical studies show both treatments are highly effective for treatment-resistant depression and rapidly reduce suicidal thoughts. However, research suggests meaningful differences in how quickly patients respond and how long improvement may last. Here are key findings from the available research:
Correia-Melo et al. (2020): This randomized, double-blind study[14] involving 63 participants showed that esketamine was non-inferior to ketamine, indicating similar efficacy in treating treatment-resistant depression.
Bahji et al. (2021): This meta-analysis[1] found IV ketamine had a 60.0% response rate and 42.6% remission rate, compared to 57.4% response and 26.7% remission for esketamine — concluding ketamine may be more effective overall.
Singh et al. (2022): This Cambridge study[15] concluded that ketamine and Spravato showed similar response and remission rates, but IV ketamine required significantly fewer treatments compared to intranasal esketamine.
Nikayin et al. (2022): A Yale study[16] indicated ketamine had better secondary outcomes than esketamine, meaning greater improvements in depression symptoms such as mood, sleep, and appetite.
Seshadri et al. (2024): A more recent meta-analysis[17] found larger antidepressant effect sizes for IV ketamine across included studies, meaning the real-world change for patients was more drastic and noticeable. The authors note direct head-to-head trials are still needed.
Spravato long-term remission data: Long-term Spravato research shows strong results for maintaining improvement in patients who respond to treatment and continue with booster doses, with nearly half of continuing patients remaining in remission at roughly two years.[2]
Does Spravato Work for Pain Like Ketamine?
No — Spravato is not used for pain, while IV ketamine has substantial evidence for treating acute and chronic pain.
- A 2019 systematic review[18] of randomized controlled trials concluded that ketamine provides significant short-term pain relief.
- A 2014 study[19] showed that low-dose ketamine produces potent relief for neuropathic pain states during administration, with some effects lasting up to three months following infusion.
- The American Society of Hematology[20] reported that ketamine worked well in patients with severe pain not responsive to standard opioid analgesics, such as sickle cell disease and cancer-related pain.
Spravato Cost vs IV Ketamine
Spravato is significantly less expensive than IV ketamine for patients with commercial health insurance. For cash-paying or uninsured patients, generic IV ketamine infusions are often the more cost-effective option.
- Insurance landscape for Spravato: Commercial plans widely cover Spravato treatments with prior approval, because the medication is FDA-approved for treatment-resistant depression. Out-of-pocket costs can be as low as $10 per session for eligible patients.
- Financial reality of IV ketamine: Health insurance carriers generally do not cover IV ketamine costs for mental health, so patients typically pay out of pocket — usually $400 to $600 per session.[3]
Uninsured individuals face a different math problem. Without coverage, a single unsubsidized retail dose of Spravato nasal spray can cost $800 to $1,200 per clinic visit — up to 6x the cost[21] of IV ketamine, making generic IV ketamine the cheaper option for cash-paying patients.
Insurance & Payment Options at Avesta Ketamine and Wellness
Avesta Ketamine and Wellness provides comprehensive insurance support for FDA-approved Spravato treatments and flexible payment and reimbursement options for out-of-pocket IV ketamine infusion costs across the Washington DC, Maryland, and Virginia (DMV) region.
Coverage and reimbursement amounts vary based on whether the patient’s provider is fully in-network or utilizes a direct billing setup. Avesta’s insurance partners include:
- In-network: Cigna
- Direct billing: United Healthcare, Aetna, Kaiser Permanente, UMR, VA U.S. Department of Veterans Affairs
The Bottom Line: Which Should You Choose for Depression?
Consider IV ketamine if you need the fastest possible symptom relief, have already tried multiple antidepressants without success, want flexibility in dosing, or are uninsured. Consider Spravato if you want stronger insurance coverage, prefer a non-invasive nasal spray, and are comfortable committing to regular clinic visits early on for relapse prevention.
Your level of symptom improvement during the first several sessions often gives the clearest answer about which option fits your depression best.
Avesta Ketamine and Wellness offers both protocols at our Bethesda, Columbia, Washington DC, Tysons, and Norfolk locations — book a free consultation to discuss which is right for you.

FAQs
Can I switch from IV ketamine to Spravato (or vice versa)?
Yes. Some patients transition from IV ketamine to Spravato for insurance or maintenance reasons. Others move from Spravato to IV ketamine if they need a different dosing approach or stronger symptom relief.
Is IV ketamine much stronger than Spravato?
IV ketamine may feel stronger or more immersive for some people because it delivers the substance directly into the bloodstream. It can also produce more noticeable short-term antidepressant effects than intranasal esketamine for certain patients. A stronger experience does not automatically translate into better outcomes, and clinicians still cannot reliably predict which treatment an individual patient will respond to best.
How long does a Spravato session last?
A standard Spravato appointment requires a two-hour in-office observation window. The FDA mandates this monitoring timeline so medical professionals can track blood pressure, manage short-term dissociative side effects, and ensure safety before clearing the patient to go home.
Do I need to take an oral antidepressant with Spravato?
Not always. In 2025, the FDA updated its guidelines to allow Spravato as a standalone therapy (monotherapy) for treatment-resistant depression. However, if you are being treated for major depression with acute suicidal thoughts, you must still use the nasal spray alongside a newly started or optimized oral antidepressant.
References
- Bahji, A., Vazquez, G. H., & Zarate, C. A. “Comparative efficacy of racemic ketamine and esketamine for depression: A systematic review and meta-analysis.” Journal of Affective Disorders, 2021. Accessed May 27, 2026.
- Wajs, E., et al. “Safety and efficacy with esketamine in treatment-resistant depression: long-term extension study.” International Journal of Neuropsychopharmacology, 2025. Accessed May 27, 2026.
- Avesta Ketamine and Wellness. “How Much Is IV and Nasal Ketamine Therapy?” Avesta Ketamine and Wellness, 2026. Accessed May 27, 2026.
- Wilkinson, S. T., et al. “The Effect of a Single Dose of Intravenous Ketamine on Suicidal Ideation: A Systematic Review and Individual Participant Data Meta-Analysis.” American Journal of Psychiatry, 2018. Accessed May 27, 2026.
- Jelen, L. A., & Stone, J. M. “Ketamine and esketamine in depression: pharmacology, efficacy, and safety differences.” Expert Opinion on Drug Safety, 2022. Accessed May 27, 2026.
- National Center for Biotechnology Information. “Glutamate and Depression.” StatPearls / NCBI Bookshelf. Accessed May 27, 2026.
- Jelen, L. A., & Stone, J. M. “Ketamine enantiomers: pharmacology and clinical use.” PubMed Central, 2024. Accessed May 27, 2026.
- Janssen Medical Information. “Comparison of Spravato and Ketamine.” Janssen Science. Accessed May 27, 2026.
- Zhang, J. C., Yao, W., & Hashimoto, K. “Arketamine, a new rapid-acting antidepressant: A historical review and future directions.” Neuropharmacology, 2020. Accessed May 27, 2026.
- Kadriu, B., et al. “Glutamatergic neurotransmission and rapid-acting antidepressants.” Discover Mental Health, 2022. Accessed May 27, 2026.
- Duman, R. S., & Aghajanian, G. K. “Synaptic plasticity and depression: New insights from stress and rapid-acting antidepressants.” Nature Medicine / PubMed Central. Accessed May 27, 2026.
- Vollenweider, F. X., et al. “Differential effects of S- and R-ketamine on regional cerebral glucose metabolism.” European Neuropsychopharmacology, 2002. Accessed May 27, 2026.
- Mathew, S. J., et al. “Mood-elevating and dissociative effects of racemic ketamine.” European Neuropsychopharmacology, 2021. Accessed May 27, 2026.
- Correia-Melo, F. S., et al. “Efficacy and safety of adjunctive therapy using esketamine or racemic ketamine in adults with treatment-resistant depression: A randomized, double-blind, non-inferiority study.” Journal of Affective Disorders, 2020. Accessed May 27, 2026.
- Singh, B., et al. “Comparative Effectiveness of Intravenous Ketamine and Intranasal Esketamine in a Real-World Setting Among Patients with Treatment-Refractory Depression.” CNS Spectrums (Cambridge), 2022. Accessed May 27, 2026.
- Nikayin, S., et al. “Anxiolytic Effect of Esketamine vs Ketamine: A Systematic Review and Meta-Analysis.” JAMA Psychiatry, 2022. Accessed May 27, 2026.
- Seshadri, A., et al. “Comparative effectiveness of ketamine and esketamine for treatment-resistant depression: A systematic review and meta-analysis.” Journal of Affective Disorders, 2024. Accessed May 27, 2026.
- Orhurhu, V., et al. “Ketamine Infusions for Chronic Pain: A Systematic Review and Meta-analysis of Randomized Controlled Trials.” Anesthesia & Analgesia, 2019. Accessed May 27, 2026.
- Niesters, M., Martini, C., & Dahan, A. “Ketamine for chronic pain: risks and benefits.” British Journal of Clinical Pharmacology, 2014. Accessed May 27, 2026.
- Brandow, A. M., et al. “American Society of Hematology 2020 guidelines for sickle cell disease: management of acute and chronic pain.” Blood Advances, 2020. Accessed May 27, 2026.
- Psychiatrist.com. “Study: Intravenous Ketamine vs Intranasal Esketamine.” Psychiatrist.com. Accessed May 27, 2026.
In this post
- How is Esketamine / Spravato® Different from Ketamine?
- How are Esketamine / Spravato® and Ketamine Similar?
- How Esketamine / Spravato® vs Ketamine is Administered
- Esketamine / Spravato® vs Ketamine: Side Effects Compared
- Esketamine / Spravato® vs Ketamine: Cost Comparison
- Is Esketamine / Spravato® or Ketamine Better for Depression?
- Does Esketamine / Spravato® Work for Pain Like Ketamine?
- The Bottom Line: Which Should You Choose?